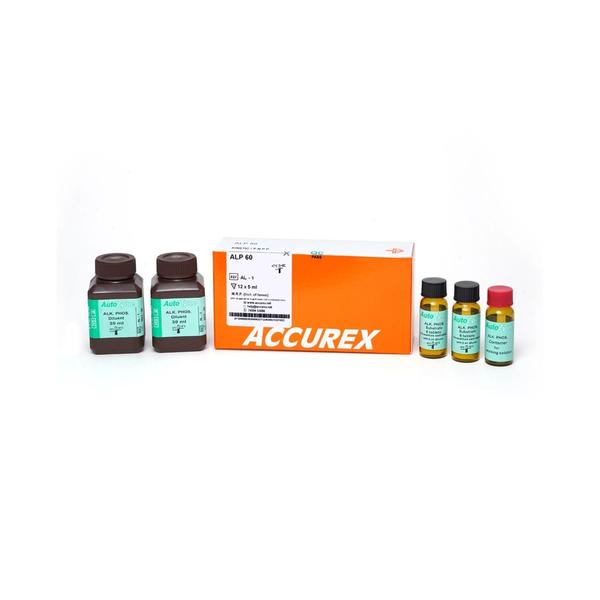
Autozyme Alkaline Phosphatase 60 Diagnostic Reagent (Pack Size : 12 x 5 ml) from Accurex. Alkaline Phosphatase 60 are used in the diagnosis of hepatobiliary disorders and bone disease associated with increased osteoblastic activity. Alkaline Phosphatase high levels may be a sign of a liver problem or a bone disorder Alkaline Phosphatase 60 reagent is set for determination of Alkaline Phosphatase activity based on kinetic method using p-nitrophenyl phosphate (p-NPP), the reagent is a Highly stable reagent & its activity can be determined in just 2.5 minutes. About Autozyme Alkaline Phosphatase is a reagent set for determination of Alkaline Phosphatase activity based on kinetic method using p-nitrophenyl phosphate (p-NPP) Autozyme Alkaline Phosphatase is a single reagent system using one step procedure Autozyme Alkaline Phosphatase has one step reconstitution. It involves dissolving substrate tablet in diluent Autozyme Alkaline Phosphatase is a Highly stable reagent Autozyme Alkaline Phosphatase activity can be determined in just 2.5 minutes Pack Size : 12 x 5 ml Principle The working solution is stablə for 21 days at 2- 8°C. The working solution should be prepared and stored in the dark (working solution bottle) provided.This is critical because the reagent is light sensitive. It should therefore be kept away from direct light. Alkaline Phosphatase cleaves p-nitrophenyl phosphate (p-NPP) into p-nitrophenol and phosphate. p-nitrophenol is a yellow color compound in alkaline medium and absorbs light at 405 nm. The rate of increase in absorbance at 405 nm. is proportional to Alkaline phosphatase activity in specimen. Quality Control To ensure adequate quality control, it is recommended that each batch should include normal and abnormal commercial reference control serum. It should be realized that the use of quality control material checks both instrument and reagent functions together. Factors which might affect the performance of this test include proper instrument function, temperature control, cleanliness of glassware and accuracy of pipetting. Other Details The kit should be stored at 2-8°C and is stable till the expiry date indicated on the label. The working solutions of Non Prostatic Acid Phosphatase & Total Acid Phosphatases are stable for 7 days at 2-8°C.
Send Message